Student Nicotine Testing Kits

Keeping Kids on the Right Path
Smoking among youth and adolescents is an issue that affects families across the country. Alcohol and substance abuse among teens remains an issue that testing can help alleviate.

Ideal for Schools and Universities
Students involved in extracurricular activities and subject to in-school drug testing reported less substance use than comparable students in schools without drug testing.

Testing Resources for Parents
One approach is for students and their parents agree to testing for drugs (and in some cases, tobacco or alcohol) on a random basis as a condition of participation in athletic or other school-sponsored activities.
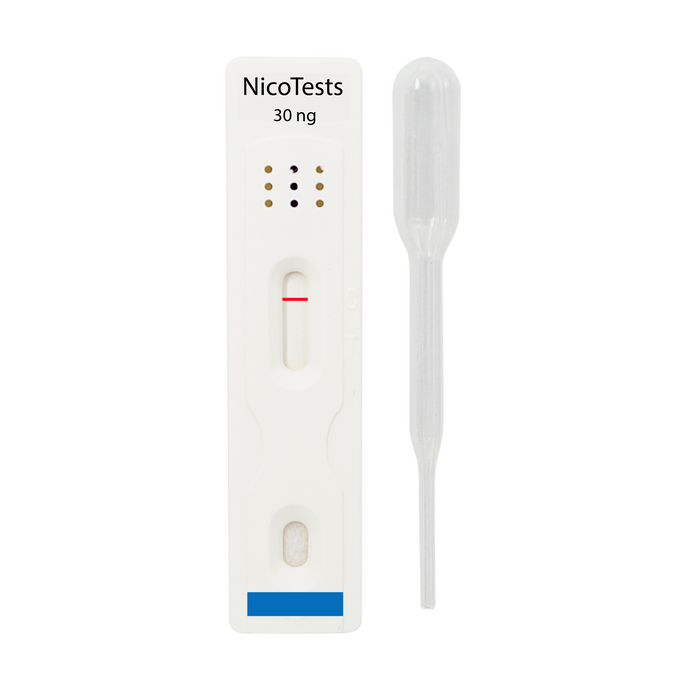
Nicotine and THC Home Test Kits
Home Urine and Saliva Drug Test Kits
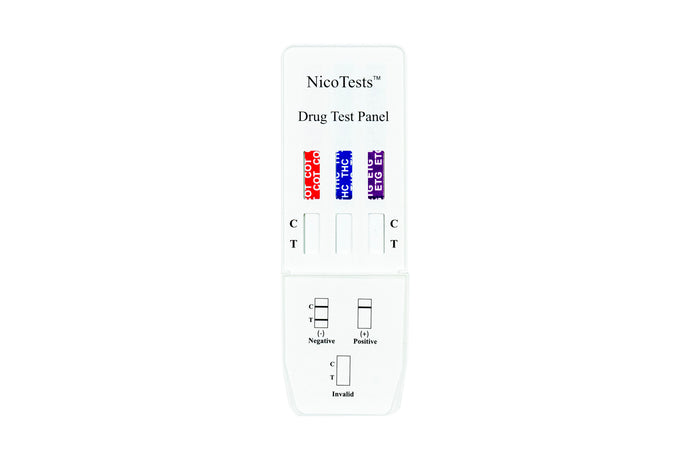
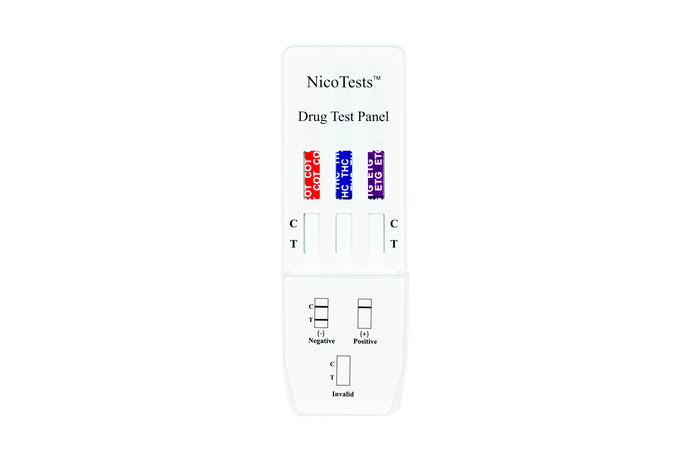
NicoTests® Home Drug Test kits
Nicotine and THC Home Drug Test Kits
NicoTests® is the leading manufacturer of saliva and urine home drug test kits. You can
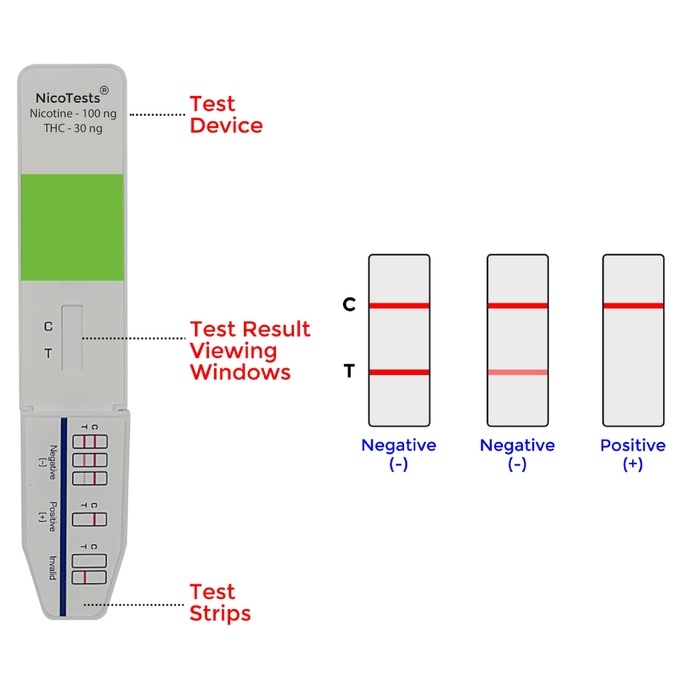
detect tobacco, vaping, cigarettes, e-cigarettes, nicotine patches, and nicotine gum use by detecting cotinine in human urine. Add Urine Temperature strips and Adulteration tests for best results. Instant drug test results in a few minutes will help you determine if your teen or workforce needs help. Protect your family and your workforce with onsite random testing with accurate drug testing by NicoTests®. Multi-panel tests can produce results for up to 16 different controlled substances. Be proactive and protect your teens.
If you are a concerned parent or social worker you can use a urine test to detect cotinine. The urine test is a good choice if a you suspect a minor has used nicotine over the last few days. An instant test provides results to parents in five to 10 minutes. Be proactive and test your teens before they become addicted to nicotine and/or other controlled substances. Testing for employees can also help prevent controlled substances like THC and alcohol from causing harm in the work place.

NicoTests® Home Drug Test Kits
Home and Office Testing solutions:
Tests for concerned parents, social workesr, family therapists, medical practices, consumers, educators, coaches, and employer job site testing. Test for up to 16 drugs with one sample. Urine and Saliva Tests for nicotine, THC, Alcohol and controlled substances. Be aware of what your teen or employee may be doing and use random testing to keep them on track.